Cleanrooms for Pharmaceutical
USP 797 Pharmacy Cleanroom
USP <797> is where most sterile compounding cleanroom conversations begin, because it ties facility design, behavior, and monitoring together.
Teams usually think in terms of the cleanroom suite: a PEC (Primary Engineering Control) supported by the surrounding rooms and airflow strategy.

Air change rates and environmental control
Working air change rates into the HVAC of your pharmacy cleanroom design helps you maintain the room after it’s been built. For example, the air change rate for a class 8 cleanroom should be 5 to 48 every hour with a 5-15% ceiling coverage. Learn more about ISO cleanroom classifications here.

Humidity requirements
Humidity matters too, especially once you’re balancing comfort, gowning, and static/particle behavior. You typically want a relative humidity of 60% or below.
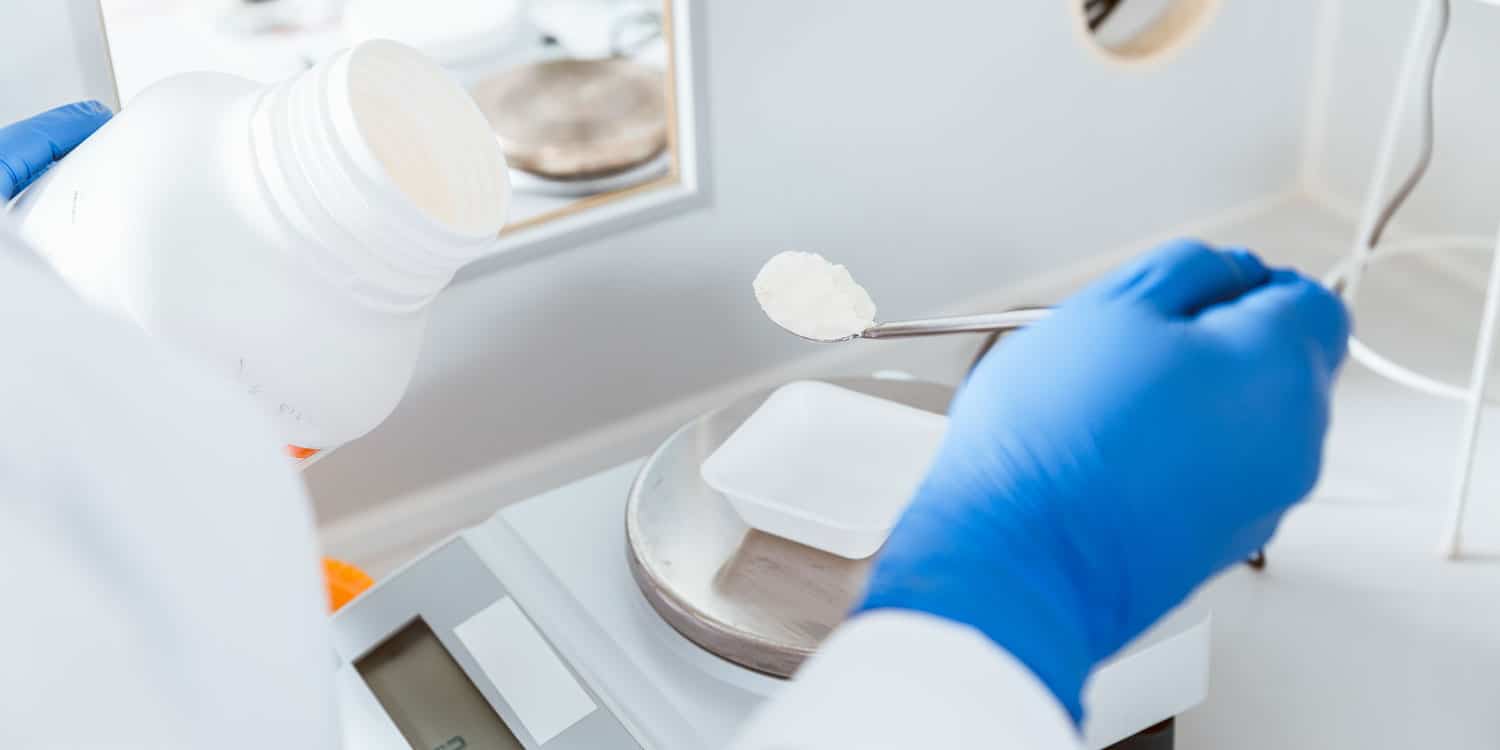
Monitoring cadence and SOP readiness
USP <797> compliance is not a one-and-done build. You’re setting up a space where certification, recertification, and environmental monitoring become routine operations.
If you’re early in planning, we recommend getting specific about what you compound and how you intend to operate. It changes everything: room adjacencies, pass-through strategy, storage, and how you stage cleaning supplies and waste streams.
Cleanrooms for Pharmaceutical
USP 800 Pharmacy Cleanroom
USP <800> is about containment. It’s built to minimize exposure risk to personnel, patients, and the environment when hazardous drugs are received, stored, prepared, and handled.
In pharmacy cleanroom design, the terms you’ll hear most often are:
- C-PEC (containment primary engineering control, the “hood” or isolator where compounding occurs)
- C-SEC (containment secondary engineering control, the room that supports the C-PEC)
- Negative pressure strategy to keep potentially contaminated air from migrating to adjacent spaces
A commonly referenced design target for negative pressure rooms is 0.01 to 0.03 inches of water column negative to adjacent areas, and a minimum of 12 ACPH for certain containment areas.
We don’t treat those numbers as decoration. They affect your door selection, pressure monitoring points, exhaust approach, and how the space behaves when people move through it.
Designed for Pharmaceutical Compounding
Most compounding teams have lived the same headache: a room can “meet the spec” on paper and still be miserable to operate in. When we design a compounding pharmacy clean room, we’re looking at the friction points that create contamination risk or slow you down.
-
Materials and Cleanability

Materials and Cleanability
Cleaning SOPs are usually one of the first considerations for pharmacy cleanrooms. Select wall, ceiling, and door materials that handle repeated disinfection and routine wear while avoiding details that trap debris or complicate wipe-down. If your team already has cleaning SOPs, we design your cleanroom suite to support them. If you’re still building those SOPs, we’ll ask the questions that your inspectors and QA team will care about later and design your cleanroom suite to set you up for success.
-
Airflow, filtration, and performance testing

Airflow, filtration, and performance testing
Cleanrooms are classified based on particle size and count per volume of air. Facilities usually use air filters to clean out contaminants such as vapor, moisture, or particulates. The air can be recirculated after cleaning, or fresh air can be pumped into the cleanroom as a replacement. Read more about cleanroom ISO classifications here.
-
Testing Considerations

Testing Considerations
Pharmacy clean room testing requirements shift depending on your operation and compliance framework. Your initial certification covers rooms and primary engineering controls. Then you’re on a recertification cycle. Particle counting shows whether you’re meeting ISO classification targets. Smoke studies are used to assess whether air moves the way it was designed to and how quickly the environment recovers after a disruption. Pressure differential testing verifies directional control, which is critical for both positive pressure sterile compounding areas (for USP 797) and negative pressure containment zones (for USP 800). Air and surface sampling frequency comes from your risk assessment and SOPs. For some operations, continuous monitoring of pressure, temperature, and humidity becomes part of the compliance documentation trail as well.
-
Pressure, separation, and entry control

Pressure, separation, and entry control
In compounding environments, pressure strategies and separation choices connect directly to workflow. People, carts, waste streams, materials, garbing steps, pass-throughs, staging. It’s all impacted. A modular pharmacy cleanroom system allows you to add or rework transitions as workflows evolve and separate processes without expanding or replacing the entire suite.
Cleaning Your Pharmacy Cleanroom
Cleaning procedures are where compliance and practicality collide. Supplies matter, storage matters, and the difference between supplies “dedicated to the cleanroom” and “borrowed from somewhere else” shows up fast in particle counts and surface sampling.
- Cleaning and disinfecting supplies should be low-lint and often disposable (wipers, sponges, pads, mop heads).
- Reusable cleaning tools should be dedicated for use in classified areas and made of cleanable materials (not porous materials like wood).
- Cleaning, disinfecting, and sporicidal agents used within the PEC must be sterile, and sterile water is required when diluting concentrated agents for use in the PEC.
When we help teams plan a room, we also talk through where those supplies live, how you keep them separated by room, and how you move them without breaking your flow.
Pharmaceutical Cleanroom Benefits
Modular USP-Compliant Cleanrooms for Any Workflow

Sterile Compounding Suite
Hospital Pharmacy
ISO 7 buffer room with dual ISO 5 laminar flow hoods, dedicated anteroom, and materials pass-through. Hardwall modular panels with seamless wall covering for repeated disinfection.
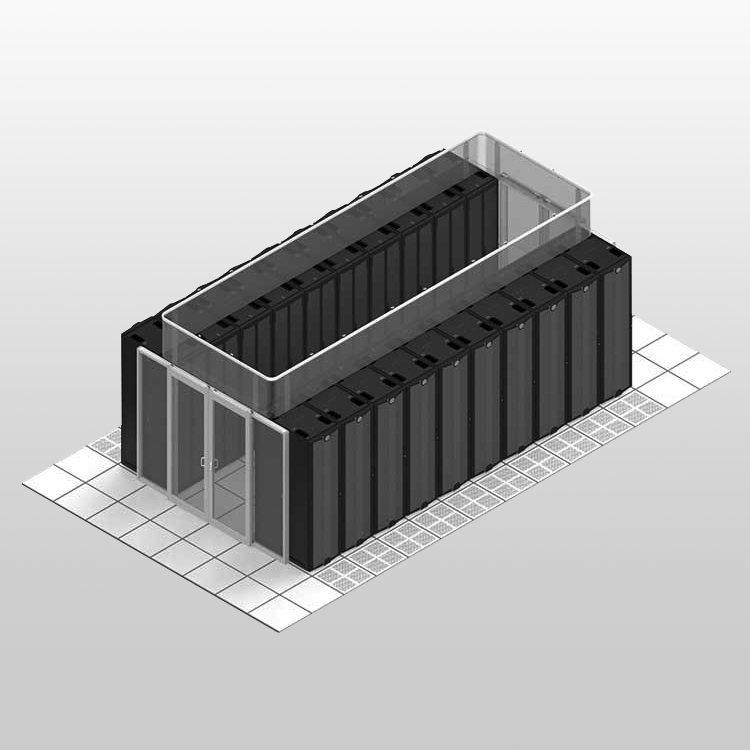
Hazardous Drug Containment
Specialty Pharmacy
Negative pressure room (-0.02 iwc) with C-PEC, ante-area, and separated gowning zone. Dual entry system with dedicated hazardous waste removal path.

Temporary Compounding Space
Regional Health System
Portable ISO 7 cleanroom installed in 48 hours during facility renovation. Hardwall panels, integrated monitoring, operational for 14-week construction period.

Workflow Expansion
Outpatient Infusion Center
Modular expansion from 240 to 320 square feet. ISO 5 hoods in perpendicular configuration. Relocated pass-through and dual gowning stations.

Integrated 797/800 Suite
Compounding Pharmacy
Combined facility with positive pressure ISO 7 sterile area and negative pressure hazardous drug room. Pressure cascade system with shared support spaces and separated workflows.
Product Categories
Our comprehensive product portfolio serves critical environments across pharmaceutical applications, including compounding. With thousands of successful installations worldwide, these modular clean room components have proven their reliability in the most demanding situations.
Trusted by Fortune 500 companies across pharmaceutical, electronics, and aerospace industries for critical contamination control.
Modular. Scalable. Trusted. Subzero Engineering.
35+ Years Combined Experience
As one of the leading prefabricated clean room manufacturers, we have engineered contamination control solutions since before modular systems became industry standard.
Proven Results
Our pharmacy cleanroom installations achieve identical contamination control performance to permanent facilities while reducing deployment time by 75%.
Global Support
In-house manufacturing across state-of-the-art facilities with local expertise through regional partners and support teams.
Consultative Approach
We start by asking “What do you want to achieve?” rather than trying to fit existing solutions to your specific requirements.
A pharmacy clean room project usually involves more stakeholders than anyone admits at kickoff: facilities, QA, pharmacy leadership, procurement, EHS, infection prevention, inspectors. Sometimes all of them.
Our job is to pull the technical and operational requirements into a buildable cleanroom plan. That consultative approach is core to how we simplify projects and avoid unnecessary complexity and delays.
Common Questions About Manufacturing Cleanrooms
What is a pharmacy cleanroom?
A pharmacy clean room is a controlled environment designed to reduce contamination risk during compounding, using filtration, airflow control, cleanable surfaces, and documented operational practices.
Our Simplex cleanrooms use HEPA filtration, fan filter units, pressure control, and access management to create contamination-controlled environments for sensitive pharmaceutical work. For technical details, see our Questions and Answers about Cleanrooms and our overview of Cleanroom Classifications and ISO Standards.
What are the rules for clean rooms in pharma?
Requirements depend on your application, the cleanliness level you need, and the compounding standards that apply to your workflow. For compounding pharmacies, USP <797> and USP <800> drive most facility decisions, along with the ISO classification framework that defines air cleanliness targets.
Our advice: define what your product or process requires before locking drawings. Our cleanroom planning guide covers the key decision points.
How do you clean a pharmaceutical cleanroom?
Cleaning starts with written SOPs, but the facility design has to support those procedures. That means flush interior surfaces and eliminating recesses where contamination can accumulate.
Our SIS 797 Hardwall Cleanroom Enclosure uses a frame design that reduces trapped areas and simplifies interior cleaning access. The reality is that more contamination comes from procedural shortcuts than from materials. Documented procedures and staff training matter as much as the room itself.
What is a cGMP pharmacy cleanroom?
A cGMP cleanroom operates within a broader quality system: documented procedures, controlled conditions, and the ability to prove performance through records and testing.
From a facility perspective, the question is what your regulator or customer expects your environment and controls to demonstrate, and what evidence you’ll need during audits. For FDA’s official perspective, see their overview of Current Good Manufacturing Practice (CGMP).
What kind of testing is most common for pharmacy cleanrooms?
Testing addresses two questions: does the room meet its classification, and does it perform when you’re working in it?
Certification is handled by qualified third parties and includes airborne particle counts, airflow velocity measurements, and volume verification. We use CFD during design to predict airflow patterns, identify turbulence, and validate air change performance against ISO targets.
What’s the difference between a USP 797 and a USP 800 cleanroom?
USP <797> focuses on sterile compounding: protecting the preparation and controlling the compounding environment. USP <800> focuses on hazardous drugs: protecting personnel and the surrounding environment from exposure. That’s why negative-pressure containment details appear in USP <800> designs.
We break down both standards and how they relate on our USP 797 and USP 800 overview, and we answer common design questions in our article USP 800: 5 Things You Need to Know.
What’s the difference between a cleanroom and a buffer room?
A cleanroom is the overall controlled environment: the full suite with its airflow, filtration, and pressure strategy. A buffer room is one room inside that suite, specifically the main ISO-controlled space where compounding occurs. The anteroom supports gowning and material transfer.
For more on how these rooms work together, see our USP 797 and USP 800 page.
Modular Clean Room Applications
Subzero Engineering’s Simplex modular clean rooms were developed with custom applications in mind. The possibilities are endless. Whatever the unique requirement, our team brings industry expertise and a proactive approach to solving even the most difficult isolation challenges.
-
Aerospace + Defense


Aerospace + Defense
We understand the complex nature of the aerospace and defense manufacturing industry.
Learn More -
ELECTRONICS MANUFACTURING


ELECTRONICS MANUFACTURING
Modular enclosure solutions for high sensitivity electronics manufacturing environments.
Learn More -
E-LIQUID MANUFACTURING


E-LIQUID MANUFACTURING
We engineer custom e-liquid, e-cigarette, e-juice manufacturing cleanrooms compliant with current FDA regulations.
Learn More -
FOOD PREPARATION


FOOD PREPARATION
Ensure a contamination free environment to process, prep, and package food.
Learn More -
HAZMAT HANDLING


HAZMAT HANDLING
Simplex cleanrooms can be custom designed to deal with the complex handling and storage of hazardous materials.
Learn More -
LASER MANUFACTURING

LASER MANUFACTURING
Airborne contaminants can wreak havoc on lasers; Safeguard your critical laser environment.
Learn More -
Manufacturing Applications


Manufacturing Applications
Simplex cleanrooms and separation products create a variety of solutions for manufacturing facilities.
Learn More -
MEDICAL APPLICATIONS


MEDICAL APPLICATIONS
Simplex cleanrooms can be designed to efficiently Meet and Exceed Industry Regulatory Requirements.
Learn More -
PHARMECEUTICAL COMPOUNDING


PHARMECEUTICAL COMPOUNDING
We have designed numerous cleanrooms for use in the pharmaceutical industry.
Learn More -
TEMPORARY & PORTABLE

TEMPORARY & PORTABLE
Temporary/portable cleanrooms can be designed using hardwall or softwall configurations and can quickly be expanded or relocated.
Learn More











